The IQ, OQ, PQ and Their Impact on Quality Control

Some constants cannot be ignored, even between two radically different products. It’s then that IQ, OQ, and PQ enter the picture!
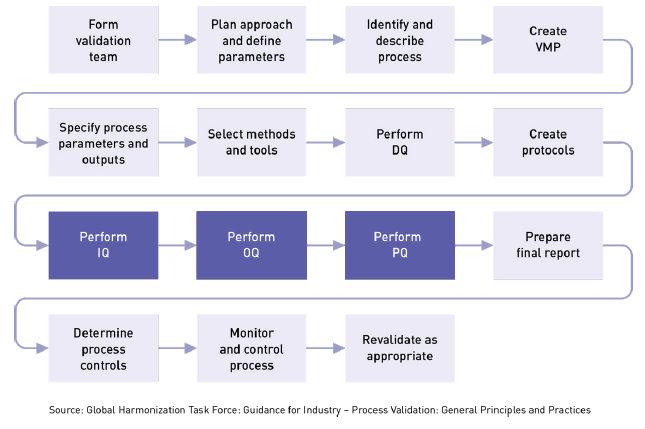
Overview of Medical Device Process Validation: IQ, OQ, and PQ – Oriel STAT A MATRIX – ELIQUENT Life Sciences Blog
A Basic Guide to IQ, OQ, PQ in FDA-Regulated Industries

LIMS Validation: What is IQ, OQ, PQ? - LabCollector Blog

Why IQ/OQ/PQ Packages Matter to Process Services

What Are IQ OQ PQ, The 3 Q's Of Software Validation Process
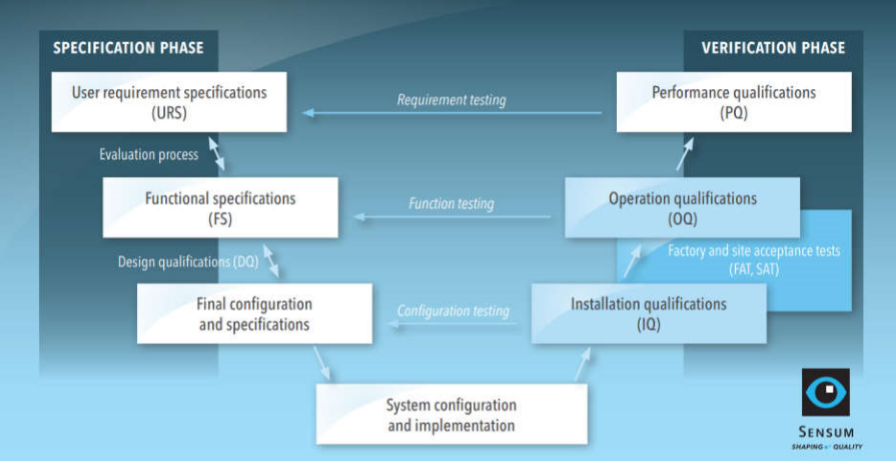
Pharmaceutical qualification and validation: tips to get through nightmares

Validation (IQ/OQ/PQ) Simplified - Forum Plastics

Writing Compliant IQOQPQ Protocols — Meeting FDA Expectations

Performance Qualification (PQ) of Pharmaceutical Equipment : Pharmaguideline
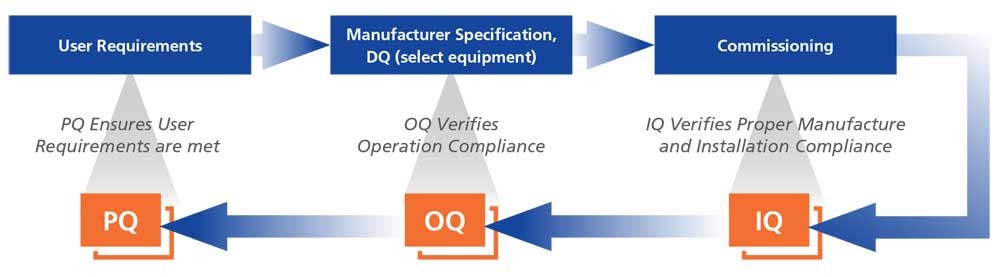
What Are DQ, IQ, OQ, and PQ, and Why Are They Required in Medical Device Industry?
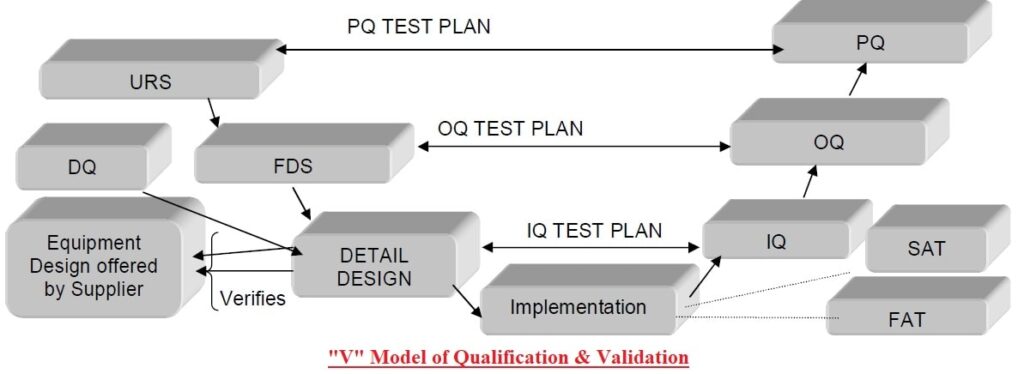
Guideline for Equipment and System Qualification - Pharma Beginners

Nathan Roman 📈 on LinkedIn: #validation #temperaturemapping #qualification

Equipment Qualification - IQ, OQ, PQ Protocols : Compliance Training Webinar (Online Seminar)









